In recent years, there has been a growing emphasis on the surgical management of endometriosis by fellowship-trained surgeons, including newly graduated surgeons, in addressing complex cases of deep infiltrating endometriosis. This includes conditions such as bilateral pelvic sidewall involvement, complete obliteration of the posterior cul-de-sac, and ovarian remnant syndrome—which can lead to ureteral torsion or difficult visualization18,19. The identification of ureters is crucial for safely managing advanced-stage endometriosis and ovarian remnant syndrome. However, this task remains challenging, particularly in cases with severe adhesive disease due to the possible alterations in anatomic location of the ureters6,20. Additionally, the excision of endometriosis around the ureters is complicated by factors such as diminished tactile feedback during robot-assisted surgery21, tissue inflammation or fibrosis, limited visualization of ureteral anatomy, and deep adhesions to the urinary tract22. In a related study, but in performing a parametrectomy, Ianieri, et.al. demonstrated a standardized approach for comparing complications and functional outcomes. Parametrectomy, even when performed by expert surgeons, carries a notable risk of bladder voiding deficits23.
Exacoustos et.al. strongly advocates for the routine use of preoperative sonographic mapping in cases of suspected deep infiltrating endometriosis (DIE). Proper patient counseling based in conjunction with imaging is not only essential for informed consent but also facilitates optimal surgical planning. This also allows for preparation of a multi-disciplinary approach for complex cases, involving general surgeons and/or urologists24. Regardless of surgeon team, ureteral mapping is a paramount part in safely performing complex surgery. A recent systematic review evaluated laparoscopic and robotic-assisted techniques for ureteral reimplantation in the treatment of ureteral endometriosis, concluding that both approaches are safe and effective25. The integration of NIRF with ICG dye has significantly expanded its application across various surgical procedures aiding in precise ureteral mapping13,15,16,17. Numerous studies have demonstrated the effectiveness of ICG in facilitating endometriosis resection, sentinel lymph node mapping, colorectal surgeries, and evaluating vascular patterns in rectosigmoid endometriosis26,27,28,29. The use of fluorescence-guided imaging in robot-assisted ureterolysis has shown potential to enhance procedural efficiency and safety11.
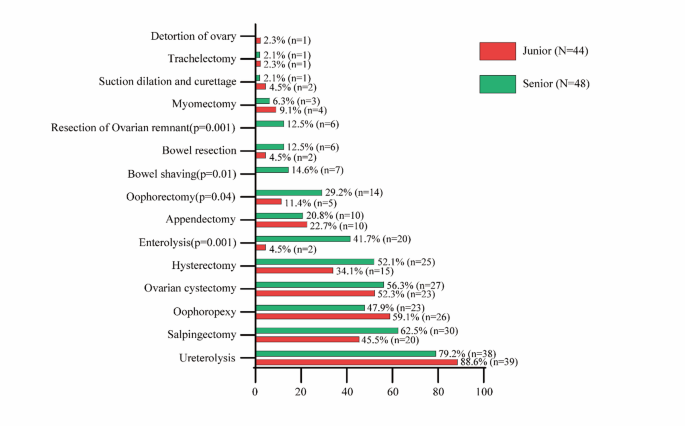
Moreover, resecting deep infiltrative endometriosis is associated with heightened surgeon anxiety. Surgical complexity is closely linked to situational stress and increased mental and physical demands, all of which contribute significantly to overall workload5. This study did not, however, address differences in stress felt between junior and senior surgeons. Our findings indicate that both groups of surgeons achieved similar surgical outcomes with comparable complication rates. Although the senior surgeon performed more Stage IV cases with ICG compared to the junior surgeons—likely due to patient selection—both groups exhibited similar complication rates. Notably, the ureterolysis rates in both groups were high (junior group: 88.6% vs. senior group: 79.2%), and no instances of temporary or permanent ureteral injury were observed in either the junior or senior surgeon groups when ICG-assisted ureteral mapping was utilized. This suggests that with ICG fluorescence for ureteral identification, even junior surgeons were confident enough to perform complex endometriosis excision without causing ureteral injury. These findings indicate that the implementation of ICG ureteral visualization may help reduce junior surgeons’ anxiety and improve safety when performing complex cases.
Regarding total operative time, the median operative times were 233 min in the junior group and 348 min in the senior group. The longer operative time in the senior group is likely due to the higher rate of patients with complete cul-de-sac obliteration, a history of abdominal surgery, and a greater number of procedures performed, such as enterolysis, ovarian remnant resection, oophorectomy, and bowel shaving. This was supported by the absence of a significant difference in total operative time after adjusting for these variables, suggesting that junior surgeons can achieve similar surgical outcomes as senior surgeons under ideal conditions. However, we acknowledge that the inclusion of concurrent procedures (e.g., enterolysis, bowel shaving) may act as confounding variables, as we were unable to obtain the exact console time for each individual procedure. Therefore, using the percentage of concurrent procedures can only partially explain differences in total operative time and cannot provide an exact measure.
Our study provides valuable insights into the use of ICG fluorescence for ureteral identification in robot-assisted surgery for advanced endometriosis. However, several limitations should be acknowledged. First, the retrospective nature of the study introduces several inherent limitations. These include: (1) the lack of randomization, which may result in selection bias, as patient assignment to surgeon groups was not controlled and could have been influenced by factors such as case complexity or surgeon availability; and (2) reliance on existing medical records, which may lead to incomplete or inconsistent data—particularly for variables that were not routinely or uniformly documented, such as analgesic usage. Second, our follow-up period was limited to six weeks post-surgery, during which no adverse effects related to ICG were observed. However, the potential long-term safety and efficacy of ICG use in endometriosis surgery remain unexplored, warranting further prospective, long-term studies to evaluate its sustained impact on surgical outcomes and patient safety.
Despite these limitations, our study has several notable strengths. This is the first study to compare the utilization of fluorescence-guided techniques by surgeons with different experience in RALS for advanced-stage endometriosis cases. This real-time surgical adjunct enhances precision and safety, particularly in cases of deep infiltrative endometriosis and severe pelvic adhesions, even for junior surgeons. A previous study similarly reported the use of endovenous ICG as an effective tool for real-time assessment of bowel perfusion during rectosigmoid resection in patients with deep endometriosis, thereby improving surgical precision and anastomotic safety30. Furthermore, our findings contribute to the growing body of evidence supporting ICG-assisted surgery, offering a foundation for future prospective, multicenter studies to validate its widespread clinical adoption. By sharing our experience, we aim to enhance surgical strategies and optimize patient outcomes in the field of minimally invasive gynecologic surgery.
The integration of ICG fluorescence into surgical workflows represents a significant advancement in robot-assisted ureterolysis, enhancing precision and minimizing risks in cases of deep-infiltrating endometriosis and ovarian remnant syndrome. Our study found no significant differences in complication rates or operative time between junior and senior surgeons, highlighting the potential of ICG-assisted ureteral mapping to shorten the learning curve for graduating fellows and improve the safety of complex endometriosis surgeries, benefiting both surgeons and patients.
For surgeons, the immediate and enhanced visualization of ICG-dyed ureters improves spatial awareness and supports surgical decision-making by providing real-time anatomical reference points. This capability allows for precise identification of ureter structures and better assessment of surgical progress, reducing the risk of complications and ensuring more accurate removal of endometriosis implants. For patients, these advancements may lead to better surgical outcomes, shorter recovery times, and fewer complications. The combination of improved intraoperative precision and real-time feedback could elevate the overall standard of care, enhancing both safety and surgical efficacy in endometriosis excision procedures.
Additionally, this study highlights opportunities to enhance training protocols in endometriosis surgery. The real-time integration of ICG fluorescence can provide trainees with a more comprehensive understanding of ureter anatomy and surgical techniques, potentially shortening the learning curve and increasing surgical proficiency. As highlighted by a recently published study, the integration of ICG fluorescence into surgical workflows has shown significant potential in enhancing surgical precision and improving postoperative outcomes in pediatric surgery, and it is recommended to establish standardized protocols and specialized training to maximize the benefits of ICG31. However, a limitation of our study is that all junior surgeon cases included ICG guidance, and we do not have a direct comparison of their performance without ICG within this cohort. We agree that further prospective, controlled studies are needed before ICG can be universally recommended as a standard training tool.
Further large-scale, multicenter, randomized studies are needed to validate the long-term efficacy of ICG ureteral identification and support its widespread adoption in gynecologic surgery. As surgical methodologies evolve, continued research will be essential in refining this technique, optimizing patient outcomes, and establishing standardized protocols to ensure the highest level of safety and efficacy in minimally invasive gynecologic procedures.
link




More Stories
Median Arcuate Ligament Syndrome | Orlando surgeon explains signs, symptoms and treatment
Gyder Surgical announces first commercial surgical cases in the United States performed by Dr. Paul M. Lombardi, M.D., at the Tri-County Orthopedics’ Joint Replacement Institute outpatient center
Bunion Surgery Market Get Facts About Business Strategies 2026 to 2031: Medi-tech Insights