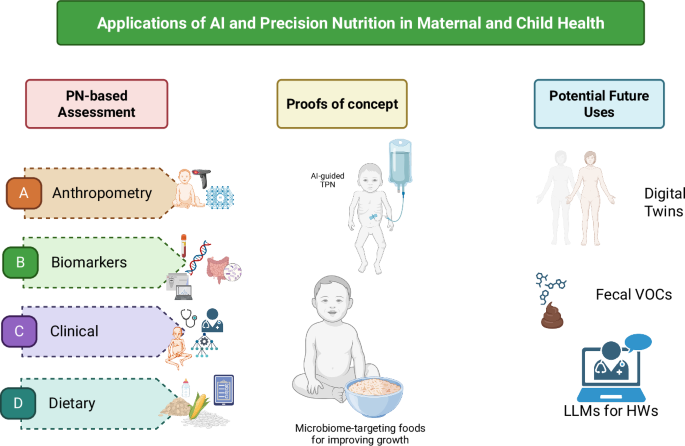
Nutritional assessment includes anthropometric measures and body composition, biochemical markers, clinical signs and symptoms, and dietary intake. These are measured and analyzed through direct measures of the body, blood collection and analysis, clinical exams, and interviews with trained personnel. Such methods have been used for decades in nutrition research and have resulted in major gains in the evidence base for nutrition and maternal and child health. However, these methods for assessment are time-, personnel- and resource-intensive, and depend on the availability of trained staff and equipment to collect and process the data, which can range from relatively simple (e.g., body weight data in a small study) to large and complex (e.g., 24-h recall data and food composition databases from repeated samples in a large cohort of mother-infant dyads)21. These methods may be complemented by integrating novel approaches to leverage increased computational power and efficiency to analyze complex multi-modal data, via artificial intelligence (AI) and machine learning (ML) models16,22. AI involves using a computer to model intelligent behavior with little human guidance23. ML is a mathematical tool that facilitates the development of algorithms to make accurate predictions from large datasets with greater accuracy than traditional statistical methods, and is of increasing interest to nutrition and health research22,24,25. Incorporating novel AI and ML methods to nutrition assessment could enable faster, more efficient, and more accurate data, translating to more accurate models and findings and inform the development and monitoring of nutritional interventions, including for maternal and child health.
Anthropometry and body composition
Established methods for nutritional assessment, including anthropometry and body composition, using measurement tapes, length/height boards, and skinfold calipers, are time-intensive and challenging to perform; require trained personnel (and may have high level of inter- and intra-rater variability in measurements); and do not automatically store data digitally, necessitating an additional data entry step26,27,28. For body composition assessment, gold standard methods include dual-energy X-ray absorptiometry (DXA), densitometry, or air displacement plethysmography (bioelectric impedance analysis (BIA),reviewed in refs. 29,30 may not be suitable for pregnant women or children), and require equipment that is costly to purchase and maintain controlled conditions, and may be less feasible in field or resource-limited settings31.
As optical imaging devices have become relatively inexpensive over the past few decades, interest in digital or automated measures of anthropometry and body composition has increased27. Three-dimensional scanning devices can objectively and relatively quickly measure the body, process the acquired data, and calculate circumferences, volumes, lengths, and surface areas27, although estimation of body composition measures (e.g., fat mass, fat free mass) from this data is more challenging32. A 3-dimensional (3D) scanner can complete hundreds of anthropometry measures in seconds32, though 3D imaging scanners vary in portability33. Smartphone and mobile phone-based technologies have advanced such that automated optical scanning systems33 and BIA34,35,36 may be captured via mobile phone, in addition to 2D images taken by the phone’s camera. These portable methods could be developed and validated for data collection in field settings at the point of need30.
Machine learning approaches are well-suited to process data from images from 3D scanners or camera-enabled mobile devices to estimate anthropometry and body composition given that (a) image data analysis can be automated reducing personnel time required; and (b) the algorithm “learns” or becomes trained and accuracy is increased as additional curated data are ingested37. In addition, taking photos of participants is faster and less labor-intensive than anthropometry or body composition measurements, with contactless data collection. For example, ML methods have been used to predict body height from single-depth images in adults by researchers such as Lokshin and colleagues38 and in multiple studies39,40. In children, height measurements and predictions can be used to detect stunting; a 2021 study in India found that a convolutional neural network-based method accurately predicted the height of standing children under 5 years of age within an acceptable 1.4 cm range among 70% of depth images, which were generated from videos from captured on a commercial 2016 smartphone; however, details on the degree of inaccuracy of the remaining 30% of depth images was not reported41. However, image and video quality are key for accurate modeling; indeed, videos with noisy data (e.g., blurry, dark, lack of the subject, or too many participants) were identified and removed from the test datasets41. In adults (n = 12 females and n = 15 males) with obesity, a 2022 study found that an automated machine learning method analyzing data from smartphone camera-enabled capture and analysis of 2D images was able to reproducibly and accurately estimate whole body fat mass compared to DXA (correlation R2 = 0.99)24, with no differences by sex. However, estimating body composition in children using image-based machine learning techniques and validating such tools in the field in pregnant women and young children remain research gaps.
Deep learning algorithms can help process images and videos but require secure server availability for app-based intake estimation for sustainability. In the future, use of convolutional neural networks or other architectures such as generative adversarial networks and deep learning algorithms will be key to process the large image-based datasets. The increased computing power helps to identify more minute details in the pictures and in the process improves accuracy42. Current methods for anthropometry and body composition assessment are constrained by high throughput. Advancing the technology to enhance reliability and reproducibility, and to optimize for individuals across the life cycle in the form of a mobile phone are important for monitoring changes in individual anthropometry or body composition over time in resource-limited settings, to develop and evaluate nutritional interventions and programs.
Biochemical
Nutritional biomarkers such as those measured in blood or urine to quantify dietary intake or nutrition status are objective and less prone to bias due to recall or reporting43. For example, minerals and vitamins can be measured in blood, stable isotopes of doubly labeled water and urine samples can enable measurement of daily total energy expenditure, and 24-h urinary nitrogen can estimate protein intake44,45. One of the main challenges in assessing nutritional status is the limited range of biomarkers that reflect intake and predict functional or clinical outcomes, such as the response to a given dietary intervention in a population. Biomarkers of nutrients and associated metabolites often reflect recent intake rather than sustained dietary habits; adding to this complexity, the metabolic rate for energy and different nutrients has been shown to have inter-individual variation46,47, possibly due to the gut microbiome18,48. In addition, some biomarkers may not accurately reflect status for nutrients that are tightly regulated, such as serum calcium or zinc49,50, in addition to changes in status prompted by inflammation (described below); finally, a limited range of nutritional biomarkers predict functional outcomes or health outcomes.
The interplay of inflammation and nutritional status may influence intra- and inter-individual variation51. The acute phase response involves the release of inflammatory cytokines such TNF-alpha, IL-1, and IL-6, which stimulate the liver to produce acute phase proteins (APP). The APPs include over 200 plasma proteins, an estimated 50% of which are involved in regulation of nutrient transport or status52. For example, serum/plasma ferritin (stored iron), retinol (vitamin A status), and zinc (zinc status) are directly affected by inflammation, both acute and chronic. In the context of acute inflammation, serum/plasma ferritin concentrations increase, whereas retinol and zinc decrease52,53. Iron trafficking may be impacted54, limiting the distribution of iron from blood to cells throughout the body in order to limit its availability to pathogens55; the liver halts the release of retinol and its binding protein49; and the transfer of zinc from blood to liver may increase50. As a result, assessment of these micronutrients without accounting for inflammation (e.g., C-reactive protein (CRP), α−1-acid glycoprotein (AGP) or other inflammatory cytokines56) may result in altered (higher or lower) micronutrient status57.
Several methods are available for population-level adjustment of ferritin and retinol, including Biomarkers Reflecting Inflammation and Nutritional Determinants of Anemia (BRINDA); however, not all micronutrients or populations are covered; BRINDA adjustment methods are available for or validated in preschool children, school-aged children, or women of reproductive age (ferritin only), but not in pregnant women or infants53,58,59,60,61. These methods use CRP and/or AGP to adjust micronutrient status to a more accurate concentration without the presence of inflammation. However, these are population-based methods for inflammation adjustment and do not typically apply to the individual level and in the context of illness. Considering that both acute and chronic inflammation (e.g., obesity and metabolic diseases) appear to impact micronutrient concentrations, accounting for inflammation as part of the comprehensive set of variables is important for precision nutrition-based strategies. These biomarkers related to metabolism (metabolites) are part of nutrient metabolism. Metabolomics, the study of these metabolites or unique fingerprints as a result of metabolic processes is an upcoming theme in nutrition research. Recently, ML methodologies such as neural networks were used to prepare an evaluation chart using nutritional biomarkers and tried to link dietary intake with biochemical profile to understand the effect on body weight62. A further challenge is to capture intra- and inter-individual variation in the metabolic and phenotypic response to a dietary intervention and ultimately predict those most likely to respond to a particular type of intervention.
Assessment of biological specimens requires central laboratories, specialized equipment, ensuring cold chain, extensive benchmarking and validation of preservation methods, and trained personnel. Methods and devices that are field-friendly (i.e., portable and not impacted by variation in environment such as temperature and humidity) and that adhere to the ASSURED criteria (i.e., Affordable, Sensitive, Specific, User-friendly, Rapid and robust, Equipment-free, Delivered)63 are particularly relevant in the context of maternal and child health and in lower-resource settings. Additionally, the availability of noninvasive methods or tests that require only small volumes of blood—particularly appropriate for populations like young children and pregnant women—is paramount to successful assessment and monitoring. Biomarker assessment at the point of care has considerable applications for screening and precision nutrition in the context of maternal and child health, particularly in resource-limited settings. For example, point-of-care assessment of vitamin A status in blood has been demonstrated, using chemical reaction that can be miniaturized in a device or test kit to facilitate use in field and community healthcare settings. Methods to screen for vitamin A status using point-of-care methods have been cataloged previously and include portable fluorometers, photometers, immunoassays, and microfluidics-based though only some were commercially available64. These devices have the potential to routinely screen for vitamin A deficiency—and evaluate interventions—particularly in settings with limited resources and infrastructure.
Gut microbiome diversity, composition, and function are also potential novel biomarkers of dietary intake, dietary quality65, nutrition status, and/or response to interventions, including analysis of fecal microbial biomarkers of food intake using AI methods66. These methods need to be implemented and validated in the context of maternal and child health, including during pregnancy, mother-infant dyads, and in children. Diversity in maternal and child populations including ethnicity, habitual diets, socioeconomic status, and age is needed for these types of studies as well as other studies such as those using gut microbiome to predict glycemic response to interventions. Validation across large, diverse populations and over time, as well as repeated analyses among similar cohorts, is needed to ensure reproducibility. In addition, evaluation of novel biomarkers compared to currently used biomarkers. For these biomarkers to accurately reflect dietary intake, further detail is needed regarding food and nutrient composition, since vitamin and mineral content in vegetables varies considerably67 depending on conditions such as season68 or post-harvest processing and storage69. Standardized approaches for biomarker validation, comprehensive and accessible food composition databases, a common ontology for dietary biomarkers, and advances in statistical procedures for novel biomarkers of dietary intake are also needed45.
Clinical
Clinical outcomes include adverse pregnancy outcomes, and metabolic diseases, including cardiovascular disease (CVD), T2DM, metabolic dysfunction-associated steatotic liver disease (MASLD, formerly known as non-alcoholic fatty liver disease (NAFLD)), obesity, hyperlipidemia, and cancers70,71. The physiological changes that occur during pregnancy and the postpartum period may unmask metabolic risk factors such as hypertension and altered glucose metabolism not observed prior to pregnancy, highlighting a key window to use AI methods to monitor risk factors and future cardiovascular outcomes72. In children and adolescents, the prevalence of obesity continues to rise, particularly in low- and middle-income settings, and is linked to persistence of obesity into adulthood and associated comorbidities and premature mortality73.
The applications of ML and AI methods in clinical examination may enable earlier intervention or treatment, particularly for nutrition-related metabolic and non-communicable diseases. Many metabolic diseases and sequelae may be assessed via medical imaging techniques, which are particularly suitable to ML and AI methods72,74,75. For example, AI-based processing and assessment of retinal images enabled early detection of retinopathy related to T2DM76. Training ML models on MRI-derived images of liver fat content along with other ‘omics and clinical data have also been used to diagnose NAFLD and outperformed existing prediction tools77. Other AI methods such as convolutional neural networks can model raw electrocardiogram signatures to detect heart rhythm dysfunctions72. They may also be useful for detecting pregnancy outcomes such as congenital anomalies and intrauterine growth retardation (IUGR)78,79.
The most accurate method for measuring low-density lipoprotein (LDL) requires beta-quantification, which is time-intensive, expensive, and infrequently used. The Friedewald equation was developed to estimate LDL using total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), and triglycerides (TG): LDL-C = TC – HDL – TGs/580. However, this equation relies on the assumptions that have not been validated in pregnancy, in children, or in the context of certain health conditions, such as HIV80. Five ML methods—linear regression, random forest, gradient boosting, support vector machine (SVM), and neural network—were used to estimate LDL-C in women with HIV (n = 5,219) or without HIV (n = 2127) compared to the Friedewald equation80. In this study, an SVM algorithm outperformed the other four ML methods and the Friedewald equation. Initial findings from this study offers support for further investigation of ML methods in predicting risk factors for metabolic health outcomes.
In a study using a Bayesian kernel machine regression (BKMR) ML approach, sex-specific differences were observed when 12 dietary components were examined in association with 10-year risk of atherosclerotic CVD81. BKMR was used to incorporate non-linear and interactive associations of dietary components with health outcomes, and account for the high degree of collinearity often observed with dietary intakes. When all other dietary components were held fixed, unprocessed red meat was associated with increased risk for atherosclerotic CVD in women. In men, fruit was non-linearly associated with lower risk atherosclerotic CVD, with an interaction between fruit consumption and whole grains was reported. BKMR identified complexities with multiple dietary intakes in association with CVD and indicate its potential in identifying which nutrient(s) or their interaction(s) are associated with disease risk by sex. Together, these methods enable more targeted precision nutrition approaches or interventions to be developed.
The potential clinical applications for machine learning in the context of precision nutrition and non-communicable diseases is evident, particularly to automate and standardize analysis of medical images. The applications of ML methods to identify risk factors or health outcomes may vary in the clinical context. For example, while there is at least one FDA-approved AI-based device available for diabetic retinopathy screening in adults, which analyzes retinal images using a cloud-based software program to output a positive or negative result82, other clinical outcomes (e.g., CVD and MASLD) are in the development stage and require validation and refinement, Further development and validation of AI and ML methods is needed for early detection of non-communicable diseases to inform early intervention and treatment.
Dietary records
Dietary intake has been identified as a modifiable determinant of individual nutritional status. Current methods for estimating dietary intake include 24-h dietary recall, food frequency questionnaire, multiple-day food records or diet records83. Although self-reported dietary intake has been evaluated using these methods in numerous epidemiological studies, measurement error, day-to-day variability, and intensive training, resources, and time burden for personnel and participants are important limitations. Food composition databases for estimating the macro- and micro-nutrient content and intake of, for example, a 24-h recall, may be limited. For example, in one study, half of the available ~100 food composition databases globally were last updated more than 10 years ago with some only available for 1980-1989, limiting the data on data such as ultra-processed foods84.
It is important to develop and validate methods for accurate dietary intake in pregnancy and during critical periods such as preconception, pregnancy, and early in life. Methods such as wearable devices85,86,87, image-based assessment88, and novel biomarkers66 may be used to accurately capture dietary intakes. For example, ML methods has been used to determine a gut microbial signature of specific whole foods (e.g., broccoli, nuts, barley) in men and women44,66. However, these methods may require additional expertise and time in processing and analyzing the resulting data88. In order to address the gap of dietary intake of adolescent females in low- and middle-income settings, a mobile AI-technology–assisted dietary assessment technique, the “Food Recognition Assistance and Nudging Insights” (FRANI) app was developed and validated against weighed food records as a ground truth in Vietnam89. Dietary intake was assessed on three non-consecutive days (i.e., 2 weekdays, 1 weekend). A smartphone with the FRANI app was provided and participants (12–18 y) were trained to take photos of each instance of food consumption. Equivalence between FRANI and weighed records was determined at the 10% bound for calories, protein, fat, and four micronutrients, and at the 15% and 20% bound for carbohydrate and several other vitamins and minerals, suggesting an accurate estimation of most intakes. Some wider bounds were observed for vitamins A and B12, possibly due to lower frequency of consumption, estimation errors, and large variance, small sample size, and limitations of FRANI in assessing vitamin A-rich fruit and vegetables and vitamin-B12–rich foods in mixed dishes. Other limitations included the need for training, recall bias, changes in eating patterns due to taking photos while eating, and limitations of recognizing less common foods. However, findings demonstrate the potential for AI-assisted dietary intake estimation in the context of maternal and child health, particularly in low-resource settings.
link




More Stories
Staff crunch cripples 64 FRUs, raising concerns over maternal and child health
Council seeks way to reduce maternal and infant health disparities
Leaders Prioritise Health Sovereignty and Women’s, Children’s and Adolescents’ Health at the 39th AU Summit